Hot melt extrusion is a process wherein active pharmaceutical ingredients (APIs) are mixed with thermoplastic polymers and other excipients, heated and mechanically sheared to form a homogeneous molten mass. This material is forced through a die and solidified into a specific shape, such as strands, films, or pellets. Originally adopted from polymer processing in the plastics industry, HME has gained strong momentum in pharmaceutical development due to its ability to improve solubility, enable controlled release, and support scalable, solvent-free manufacturing.

Low-volume hot melt extrusion (LV-HME) systems are increasingly adopted in pharmaceutical research and early development environments where material availability, flexibility, and speed are critical. Unlike production-scale extruders, low-volume HME enables efficient formulation development using minimal quantities of active pharmaceutical ingredients (APIs) while preserving the fundamental advantages of extrusion-based processing.
Hot Melt Extrusion (HME)
Hot Melt Extrusion (HME) is a continuous manufacturing process widely used in the pharmaceutical industry to enhance drug formulation and delivery.1 It is a solvent-free, continuous and scalable manufacturing process that has precise control over temperature and results in high degree of mixing and uniformity. The process starts by feeding the APIs and excipients into the extruder where the polymer is heated and sheared to disperse the API uniformly. The molten mixture is pushed through a die, cooled and shaped for downstream processing.
Low-volume HME (LV-HME) refers to extrusion processes designed to operate with small material quantities and reduced batch sizes, typically using laboratory-scale or micro-extruders. These systems replicate the thermal and mechanical conditions of larger extruders but with significantly lower material requirements. Typically, micro extruders use small barrels with precise control of temperature and shear while also monitoring process conditions and material properties.
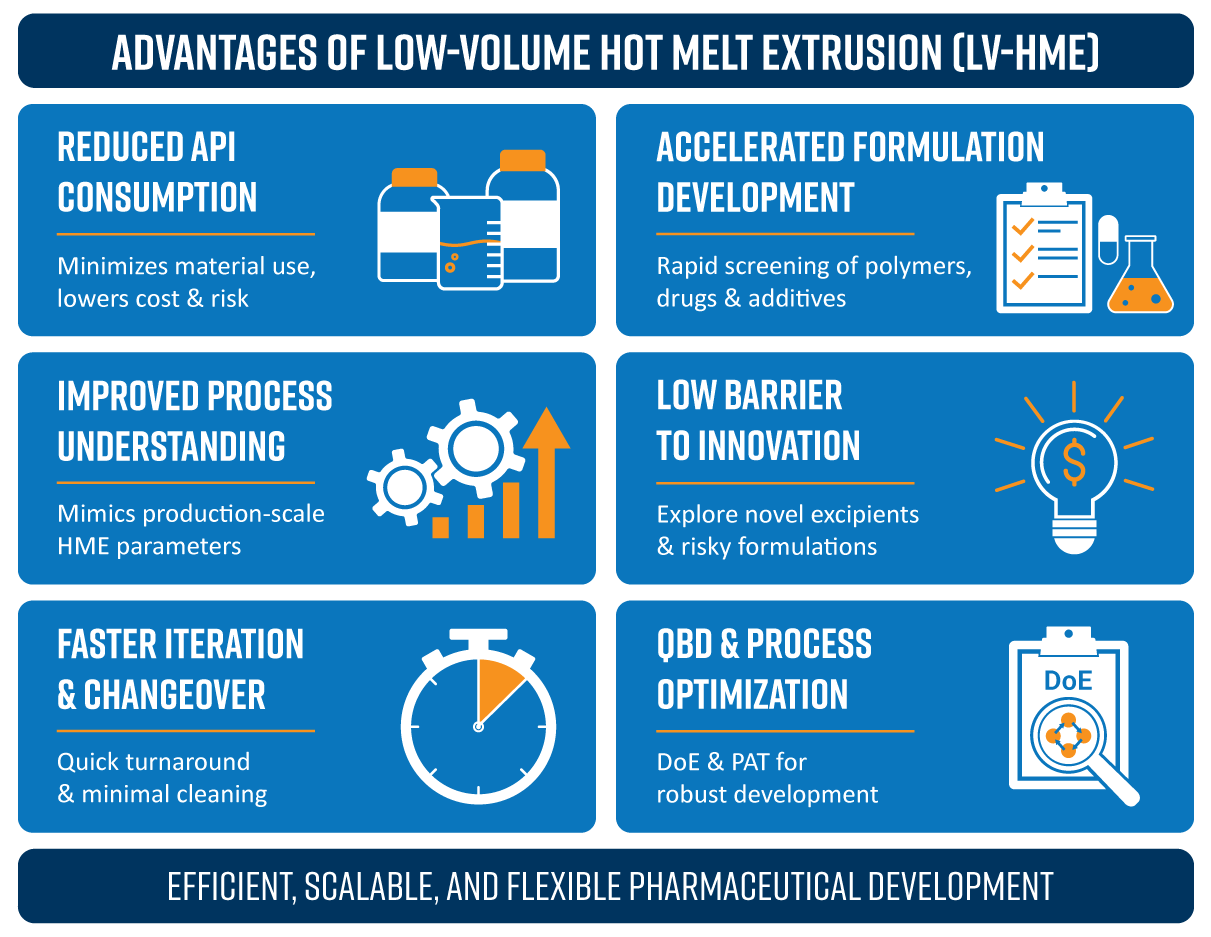
Advantages of Low-Volume Hot Melt Extrusion (LV-HME)
Reduced API Consumption
Many early-stage drug candidates are available only in limited quantities or are extremely costly. Low-volume HME minimizes material usage while still allowing realistic process evaluation. Conventional HME requires tens to hundreds of grams while low volume HME uses milligrams to few grams of APIs to enable formulation screening. This significantly lowers development cost and risk.
Accelerated Formulation Development
Low-volume extruders support rapid screening of polymer carriers, drug loadings, additives and processing temperatures. Formulations can be evaluated quickly without committing to large batches, enabling faster decision-making in early development.
Improved Process Understanding and Scalability
Low-volume extruders replicate the critical process parameters of production-scale HME, including thermal history, shear, and residence time. This allows for early identification of processing windows, development of scalable formulations and reduced risk during scale-up to commercial systems.
Lower Barrier to Innovation and Risk-Taking
Low-volume HME encourages innovation without the financial and material penalties associated with large-scale trials. Because material and operating costs are low, formulation scientists can explore higher-risk polymer systems, novel excipients and complex release profiles.
Faster Iteration and Easier Changeover
Small volume extruders require shorter start-up and shutdown times, minimal cleaning and reduced cross-contamination risk. This makes them ideal for multi-formulation labs and contract development settings where flexibility is essential.
Support for Quality by Design (QbD) driven development
Low-volume HME is well suited for QbD-driven development. It enables design-of-experiments (DoE) with minimal material, facilitates mapping of critical material attributes (CMAs) and critical process parameters (CPPs) and is compatible with inline or at-line analytical tools. This helps establish robust control strategies early in development.
Applications of Hot Melt Extrusion in Pharmaceuticals
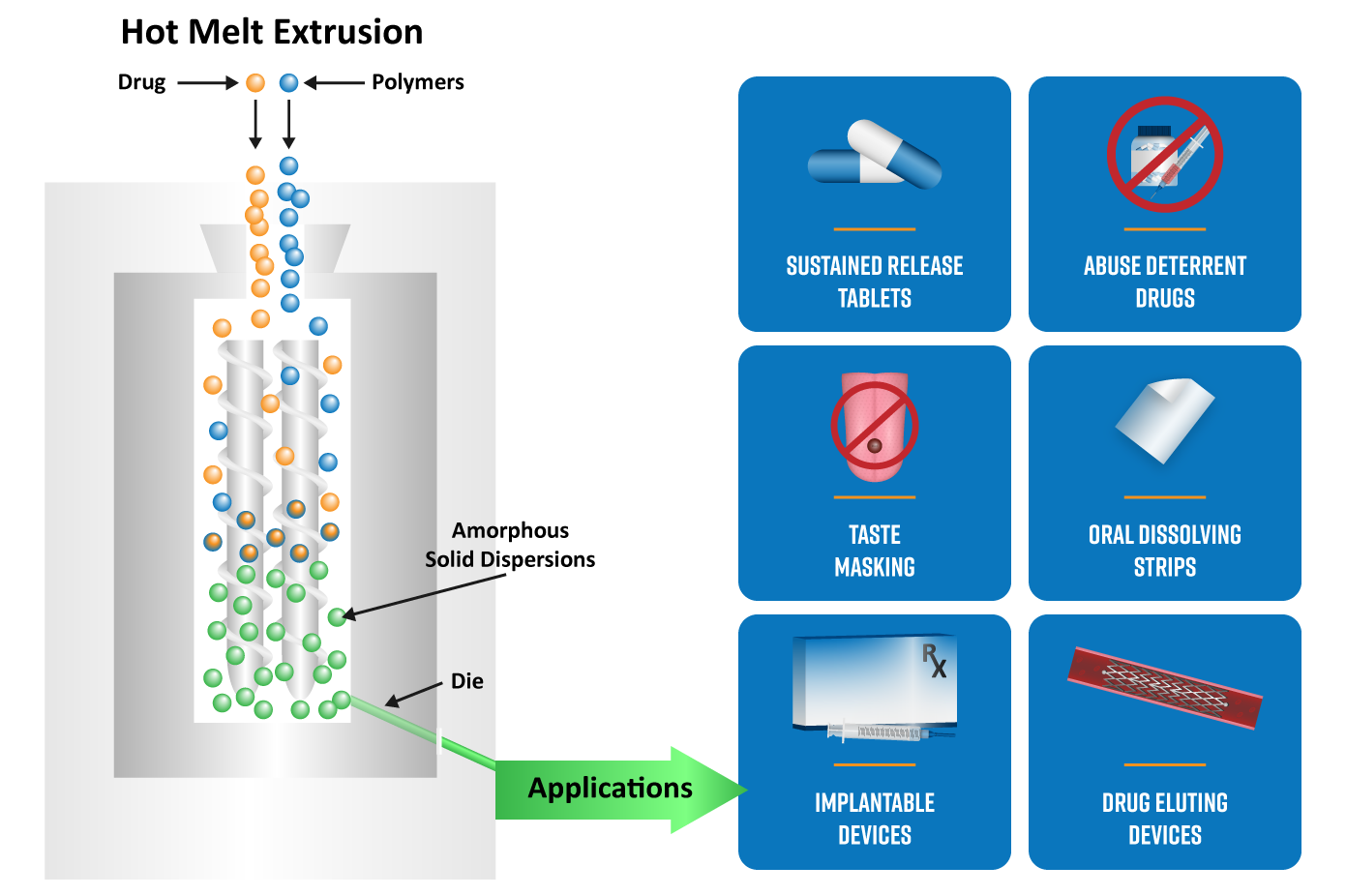
Improving Drug Solubility and Bioavailability
Many crystalline APIs exhibit low aqueous solubility and slow dissolution. HME enables the formation of amorphous solid dispersions (ASDs), where the drug is molecularly dispersed in a polymer matrix (as previously shown in Figure 1). This significantly improves dissolution rate and bioavailability compared to crystalline drug forms.2
Controlled and Modified Drug Release
By selecting appropriate polymers and extrusion conditions, HME can be used to tailor drug release profiles. The polymer matrix acts as a diffusion barrier, allowing precise control over how and when the drug is released. This can be particularly useful in developing sustained-release tablets, delayed or enteric-release formulations and abuse-deterrent formulations.
Taste Masking
HME can encapsulate bitter APIs within a polymer matrix, reducing drug exposure to taste receptors.3 This is particularly useful for pediatric and geriatric formulations, where patient compliance is critical.
Enabling Novel Dosage Forms
Extruded materials can be further processed into tablets, capsules, films and oral dissolving strips, implants and drug-eluting devices.2 This versatility supports both conventional and advanced dosage forms.
Continuous Manufacturing and Quality by Design (QbD)
HME aligns well with modern pharmaceutical manufacturing strategies by supporting continuous processing and enabling real-time process monitoring (PAT). This improves batch-to-batch consistency and reduces manufacturing waste.1 These advantages make HME attractive for regulatory-compliant, cost-efficient production.
Xplore Micro-compounders
Xplore lab-scale micro-compounders are designed for precise, efficient processing in research and development environments. They accurately replicate industrial extrusion conditions while requiring only minimal material, making them ideal for early-stage formulation and development. Xplore micro-compounders require just 5 – 40 mL of materials, enabling meaningful testing of rare, high-cost, APIs with minimal waste. Fully intermeshing twin-screw delivers strong shear and efficient distributive and dispersive mixing, ensuring uniform dispersion of API’s, additives, and excipients. The conical barrel design promotes smooth feeding, stable flow, and consistent pressure, improving mixing uniformity and process stability. A recirculation channel enables extended, well-controlled residence time, effectively simulating very long L/D ratios and many different screw elements, allowing precise optimization of shear and thermal history before extrusion.
Summary
Hot melt extrusion is a robust and versatile pharmaceutical manufacturing technology that enables improved drug solubility, controlled release, and innovative dosage forms. By combining solvent-free processing with continuous manufacturing, HME addresses key challenges in modern drug development and supports scalable, high-quality pharmaceutical production.
Low-volume hot melt extrusion offers a strategic advantage in pharmaceutical development by enabling material-efficient, rapid, and scalable formulation screening. It reduces API consumption, accelerates development timelines, supports QbD principles, and lowers the risk associated with scale-up. For early-stage drug development and advanced formulation research, low-volume HME provides a practical bridge between laboratory experimentation and commercial manufacturing.
References
- Matić, J., Paudel, A., Bauer, H., Garcia, R. a. L., Biedrzycka, K., & Khinast, J. G. . Developing HME-Based Drug Products Using Emerging Science: a Fast-Track Roadmap from Concept to Clinical Batch. AAPS PharmSciTech, 2020, 21(5), 176. https://doi.org/10.1208/s12249-020-01713-0 ↩︎
- Patil, H., Tiwari, R. V., & Repka, M. A. Hot-Melt Extrusion: from Theory to Application in Pharmaceutical Formulation. AAPS PharmSciTech, 2015, 17(1), 20–42. https://doi.org/10.1208/s12249-015-0360-7 ↩︎
- Maniruzzaman, M., Boateng, J. S., Chowdhry, B. Z., Snowden, M. J., & Douroumis, D. A review on the taste masking of bitter APIs: hot-melt extrusion (HME) evaluation. Drug Development and Industrial Pharmacy, 2013, 40(2), 145–156. https://doi.org/10.3109/03639045.2013.804833 ↩︎







