Biomaterials and nanoparticles (NPs) are used in a wide range of different health care applications from implants to dialysis membranes and cell carriers. Important for all types of biomaterial implants are their biocompatibility, i.e. how they are perceived when they come in contact with the body’s tissues and fluids. However, most biomaterials and NPs are far from ideal and induce strong adverse reactions through activation of the blood’s cascade system, i.e. the innate immune system (complement system) and the coagulation system leading to inflammation and thrombosis.
The aim of our research project is to find the connections between activation of the innate immune response and specific properties of the biomaterial surfaces and to identify what chemical and biochemical mechanisms that are responsible for the observed activation.
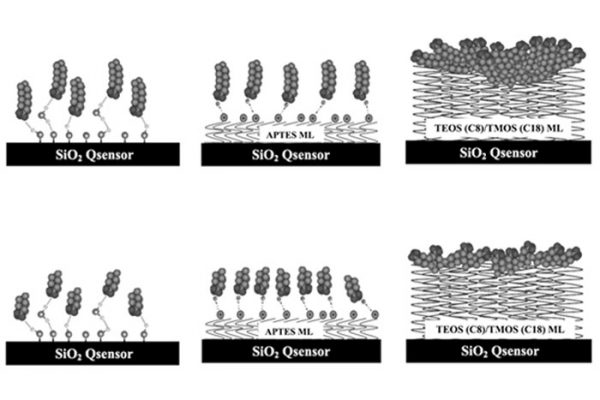
In order to investigate this type of complex and multifaceted reactions involved in the innate immune response, relevant and sufficiently complex laboratory methods are required. Our approach is to use a whole blood model, which enables us to investigate the biocompatibility of biomaterials in vitro in human whole blood in a unique way. These studies are then supplemented with plasma protein profiling and physicochemical protein analysis methods, such as QCM-D to gain a broad understanding of the mechanisms behind biomaterial-triggered contact activation of the blood’s cascade system, all the way down to molecular level. Using QCM-D, the influence of specific surface features on key initiator proteins in the coagulation and complement systems are investigated in order to identify critical conditions for the formation of specific protein-protein complexes that prompt the activation of the cascade systems. The goal is then to be able to control the properties of the biomaterial to obtain a specific biological response. In this way, a new generation of functional biomaterials can be designed, which provides the desired activation in blood and thereby greatly improves patient safety.