Battery technology continues to experience massive growth and innovation that is driven by the electrification of everything. Some of the biggest challenges faced in this industry are designing batteries with higher energy and power density, reducing charge times, extending battery lifetimes, and reducing the cost and environmental impact of manufacturing.
Nanoscience Instruments provides seven distinct solutions to scientists involved in battery research:
- Characterize surfaces and individual particles with automated SEM and EDS
- Prepare artifact-free samples for SEM using an Ion Mill
- Construct safer, improved separators with nanofiber materials made by electrospinning
- Characterize wettability using optical tensiometers
- Analyze electrolyte uptake with force tensiometers
- Monitor SEI formation in situ by pairing electrochemistry with a quartz crystal microbalance
High Resolution analysis of battery materials
The Phenom XL G2 Desktop SEM (scanning electron microscope) provides high-quality microstructural and chemical analysis for battery materials. The microscope’s small form factor requires minimal lab space, allowing the placement of the Phenom at the site of need. Combining the proven ease of use and fast workflow of the Phenom series with the largest chamber and sample stage of any desktop SEM results in a powerful tool for high-throughput analysis.
- Images are < 60 seconds away after loading the sample
- 100 x 100 mm2 sample stage – the largest of any desktop SEM
- Enhanced functionality with EDS (energy dispersive x-ray spectroscopy)
- Fits within a glove box so you can handle air-sensitive samples with confidence
- Predefined and customizable automation workflows for high-throughput analysis
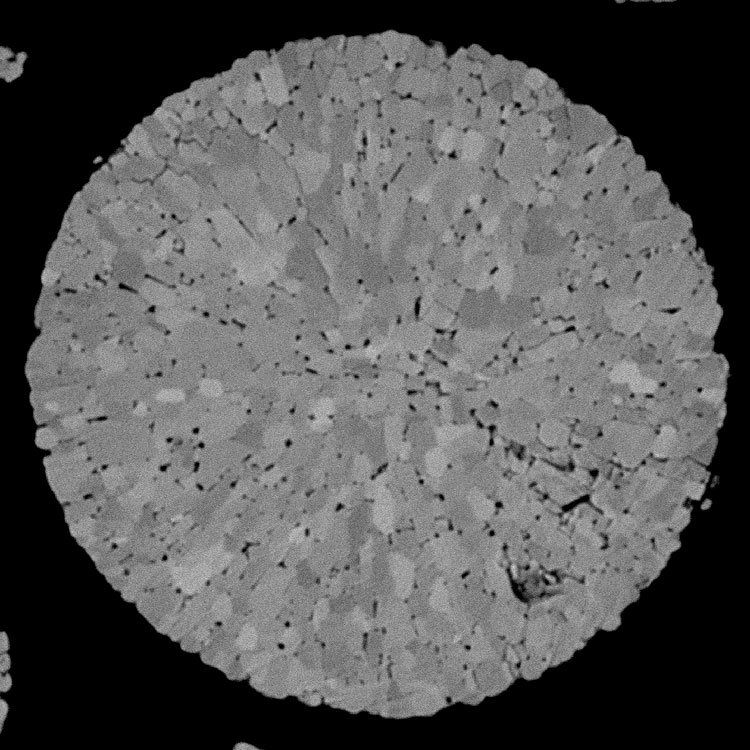
EDS Elemental Mapping


The map above shows the distribution of elements in a cathode tape cross section. EDS mapping allows for the quick separation of phase and identification of contamination.
Automated SEM Analysis of Battery materials
Size, morphology, and composition of electrode active materials are integral parameters for controlling battery performance. Automated SEM analysis is a powerful tool that can be used to analyze these properties across thousands of particles with just a few clicks.
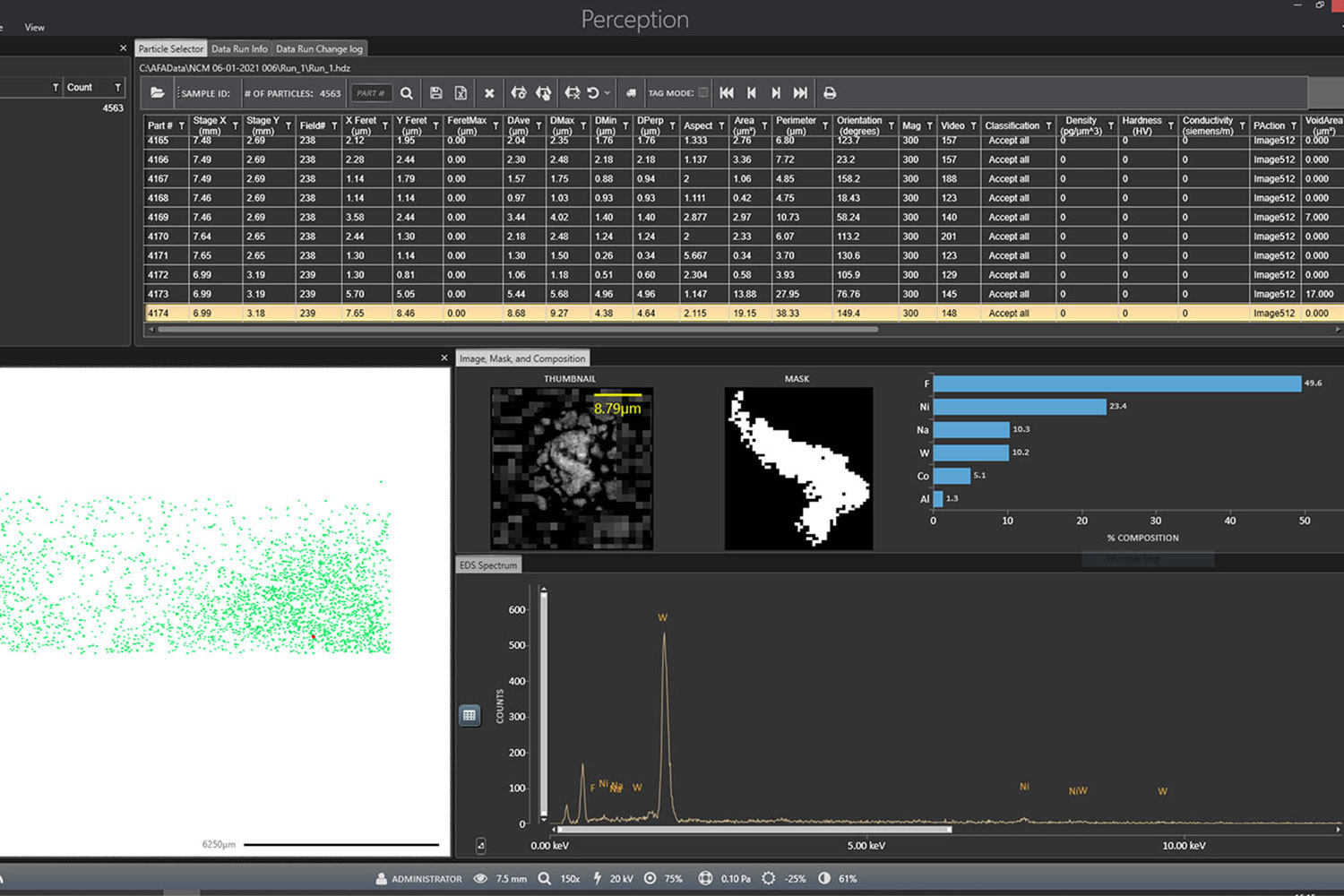
ParticleX Battery equips the Phenom XL to automatically scan samples, identify particles of interest, analyze each particle’s composition using EDS, and compiles all acquired data into templated reports for quick and easy interpretation. Typical applications include:
- Monitoring powder purity and environmental cleanliness level in production floor
- Detection and characterization of contaminants in electrode tapes
- Identification of needle-in-a-haystack “killer” particles based on automated conductivity classification
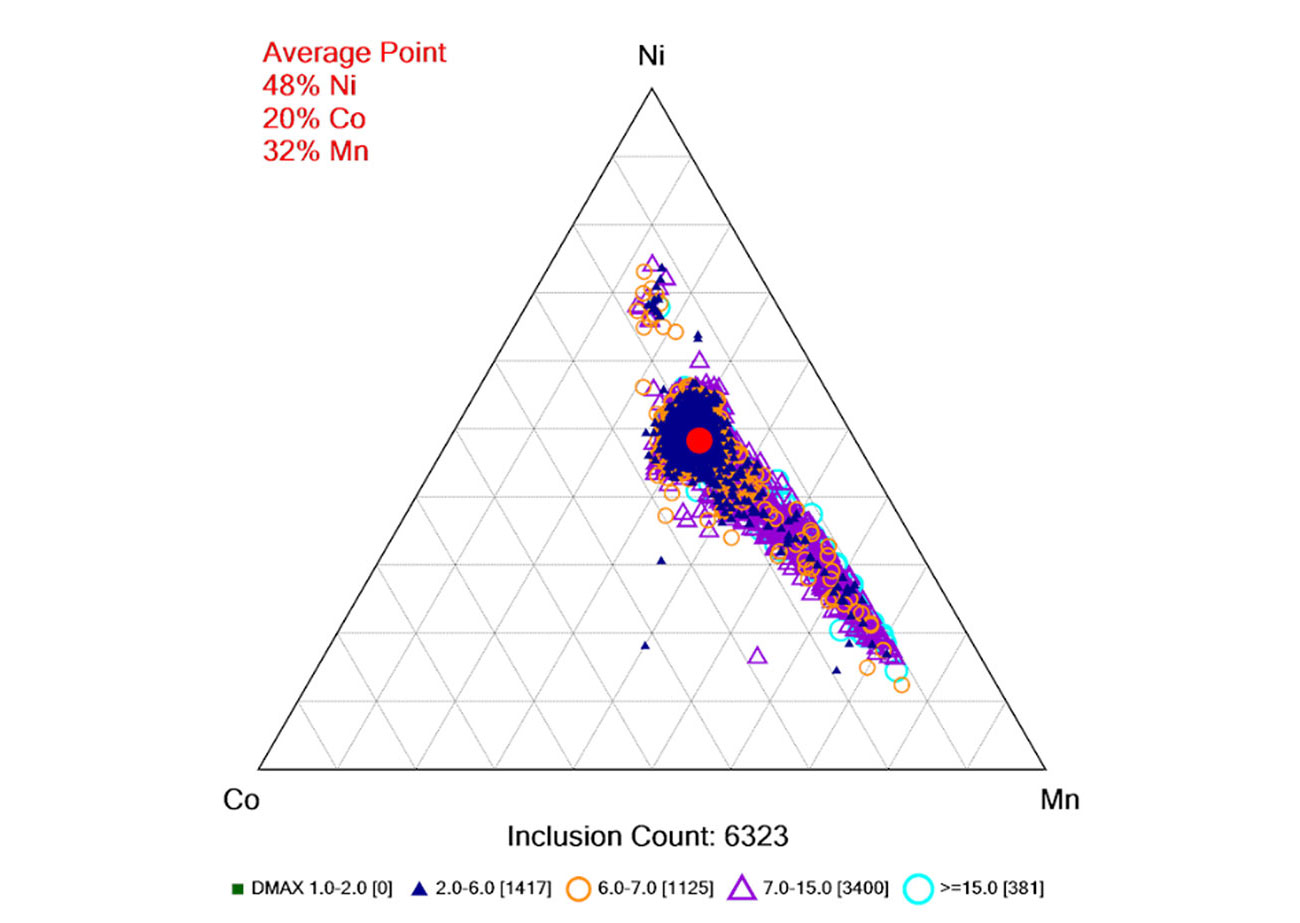
Particle-by-Particle Analysis of Cathode Active Material
ParticleX can automatically compile results into ternary diagrams showing the distribution of size and composition across a sample. In this analysis of over 6,300 NCM-523 particles, the average composition matches closely to the expected ratio of each element. The ternary diagram, however, shows a 20% fluctuation in cobalt composition, and a much larger fluctuation in the Ni/Mn ratio. Furthermore, the diagram shows that this fluctuation is more prevalent in the larger particles. This level of information can only be obtained through a particle-by-particle characterization.
Preparing artifact-free battery samples for SEM
Battery materials can be challenging to prepare for high-resolution SEM analysis. Containing several interfaces composed of thin films, powders, and organic separators, they are susceptible to damage and require a high-quality surface finish to reveal grain structures and precise layer thicknesses. Ion milling is the ideal technique for preparing such materials as it can prepare artifact-free surfaces suitable for SEM analysis.

The SEMPrep SMART meets the highest demands for surface polishing and slope cutting of battery materials.
- Largest range of ion energy (100 eV – 16 keV)
- Fully automated for unattended operation
- Vacuum transfer unit for air-sensitive samples
- Dry nitrogen venting
- Dual cooling options: Liquid N2 or Peltier
Use Cases
- 100 mm2 treated area
- Homogeneous within 10%
- 360° in-plane rotation of sample stage with adjustable direction and speed
- Versatile holders for different sample geometries
Slope cut
- Fast milling rate
- At least 1 mm wide; variable depth
- In-plane oscillation ±10° to ±120° (10° step size)
- Sample holders for 30° and 90° slope cuts with built-in titanium mask

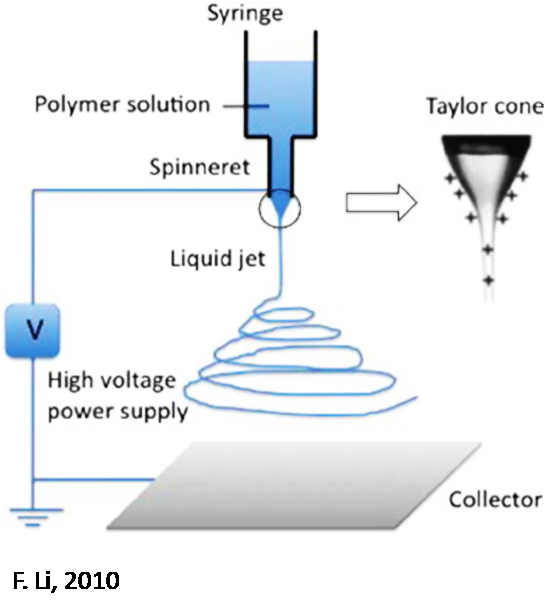
Electrospun Separators
In standard separators, porous polypropylene films or polyolefin-based membranes are employed as barriers between anodes and cathodes. However, they exhibit unfavorable characteristics in terms of battery utility such as low electron conduction, high volume expansion, structural instabilities, low wettability, and inferior thermal stability.
Electrospinning is a powerful technique used to generate nanoscale fibers that show immense promise as separators with high safety and increased efficiency compared to traditional polymers or membranes. Interconnected pores and large surface-area-volume ratios of the fibers succeed at:
- Rapid uptake of soaked electrolyte
- Diminishing degradation rates during charge and discharge processes
- Boosting ionic conductivity and the overall power response of the battery
- Increasing tensile strength through mechanical aptitude
- Maintaining thermal stability and improving conductive properties
Our suite of electrospinning equipment is designed to fabricate nanofibrous material with tunable morphologies. They are capable of generating viable structures that have the potential to be employed as efficient battery separators.
- Electrospun separators have improved wettability and ionic conductivity due to high porosity, large surface – volume ratios, and interconnected pores
- Improve thermal stability and electrochemical performance using sequential fabrication and multilayer techniques
- Flexibility of materials enables fine-tuning of separator performance: monolayer or multilayer, modified, composite, and gel polymer separators

Wettability Characterization by Optical Tensiometry
A critical step in the manufacturing of lithium-ion batteries is the pumping of the electrolyte solution into porous electrodes, with the goal of fully permeating the pores. Electrodes with poor wettability can lead to significant delays in the manufacturing process. Electrodes that insufficiently absorb the electrolyte also exhibit erratic reactions, unstable formation of the solid electrolyte interphase (SEI), and lithium dendrite formation, which all contribute to deteriorating performance and poor device life cycle.
Characterizing the wettability of the electrode material can easily be carried out by contact angle measurements at different locations on the surface of the electrode using optical tensiometry. Various contact angle measurement methods including standard sessile drops, advancing contact angles, and receding contact angles can provide important information about the electrode material’s wettability, and ultimately, its suitability for use in battery manufacturing.
The Attension Theta Flow is an automated optical tensiometry platform that leverages its advanced features to characterize surface properties of battery anodes, cathodes, and separators using contact angle and surface free energy measurements.
- Automated optical tensiometry platform with easily interchangeable modules for surface topography and picoliter-volume contact angle studies
- Enables electrolyte wettability optimization using predictive modeling and surface free energy measurements
- Plug-and-play modules controlled by pre-programmable recipes create an easy workflow
- Maintains high accuracy, traceability, and reproducibility across experiments
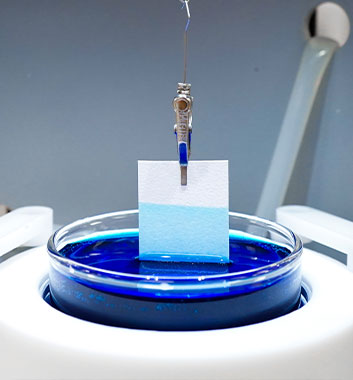
Absorption Analysis by Force Tensiometry
The separator material in lithium-ion batteries prevents short circuiting by creating a physical barrier between the two electrodes, while still allowing for lithium-ion transport. In addition to these properties, the separator material needs to absorb the electrolyte quickly during the manufacturing process. Full wetting of the separator by the electrolyte is also necessary for proper ionic transport. Therefore, a good understanding of the separator’s wettability is critical to battery development and manufacturing.
Force tensiometers enable precise characterization of the interactions between gas, liquid, and solid phases. In battery research, force tensiometers can be used to investigate the mass of electrolyte absorbed as a function of time, and kinetics for the rate of absorption. These critical measurements can help guide the development of optimized separator materials.
The Sigma 700 automated force tensiometer accurately measures mass uptake through electrolyte imbibement, wettability properties, surface and interfacial tension, dynamic contact angle and a host of other surface characteristics.
- Comprehensively study density, sedimentation, adhesion force, contact angles, and surface free energies
- Tailored for in-depth analysis of porous separator substrates
- Acquire time-dependent functions describing mass of electrolyte absorbed
- Live analysis yields rate of absorption
- Complete flexibility to study solids, liquids, and powders
- Multiple measurement options on a large range of sample sizes
Solid Electrolyte Interphase Analysis by EQCM-D
The SEI (solid electrolyte interphase) is a layer of solid salt degradation and solvent reduction products that governs reversible charging processes and overall stability in batteries. SEI accrues at the mass-exchange boundary between the anode and electrolyte and is poorly understood due to dynamic, time-dependent behavior and unclear progression.
EQCM-D (electrochemical quartz crystal microbalance with dissipation), a precise analysis technique capable of measuring changes in mass at nanogram levels, facilitates the characterization of the passivation layer formation and its structural changes in real-time. This enables researchers to quantitatively estimate delicate mass changes, SEI shear modulus, SEI viscosity, explore electro- polymerization, ion intercalation, corrosion, and electrodeposition at multiple harmonics, among other properties relevant in battery research.
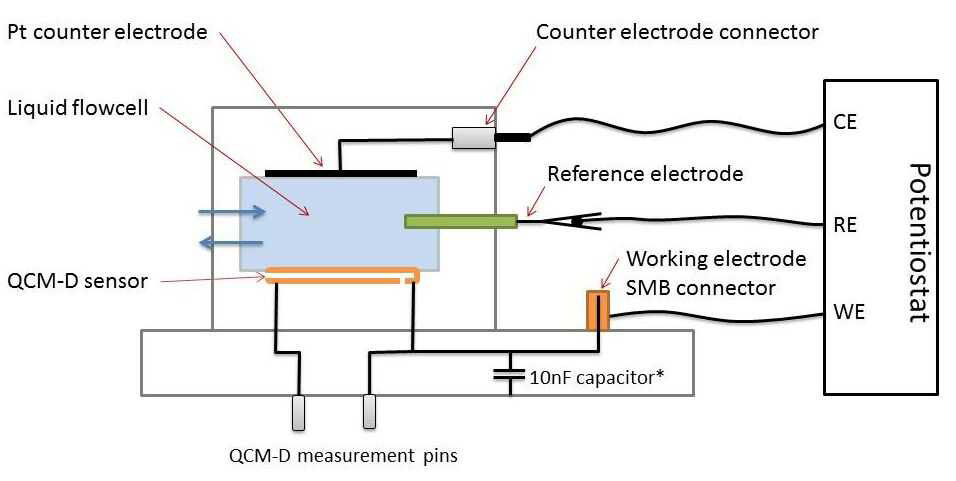
QSense EQCM-D enables real-time measurements of mass and viscoelastic properties at surfaces and can be used in the analysis of SEI alongside potential development of new electrode materials.
- In situ monitoring of dynamics and mass exchange at the SEI
- Identify conditions in which SEI is optimized for battery performance
- Easily track and manage time evolution of electrolyte boundaries in real-time

The team of scientists at Nanoscience Analytical provides our clients with expert guidance and access to the latest technologies for material and device characterization. Our scientists can:
- Perform proof of concept experiments to spur development projects
- Collect data for project justification & acquisition of new technologies
- Develop material characterization processes to exacting standards
- Support iterative device design utilizing robust analytical methods
- Evaluate a wide range of materials including polymers, composites, & ceramics
- Provide insights during failure analysis investigations

Scanning Electron Microscopy
- Microstructural analysis
- Elemental mapping
- Fiber/pore/particle analysis

Tensiometry
- Electrolyte uptake
- Separator/electrode wettability
- Absorption kinetics

Bulk Porometry
- Porosity
- Gas permeability
- Mean flow pore diameter
- Electrochemical Impedance Spectroscopy
- Electrode Capacity Measurements
- Electrode Rate Capability
- Battery Cycling Analysis